Research Grade Foravirumab
All
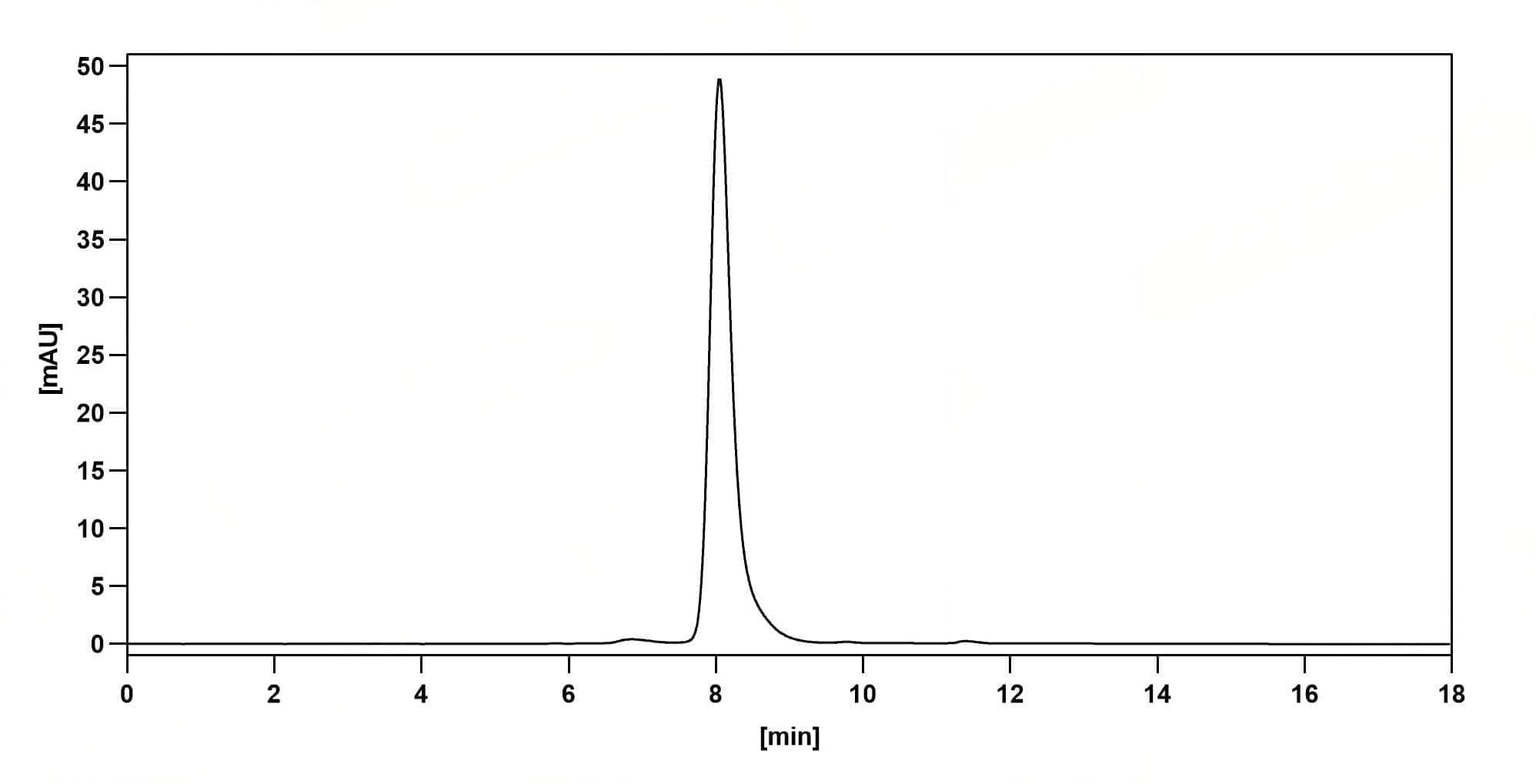
SEC-HPLC
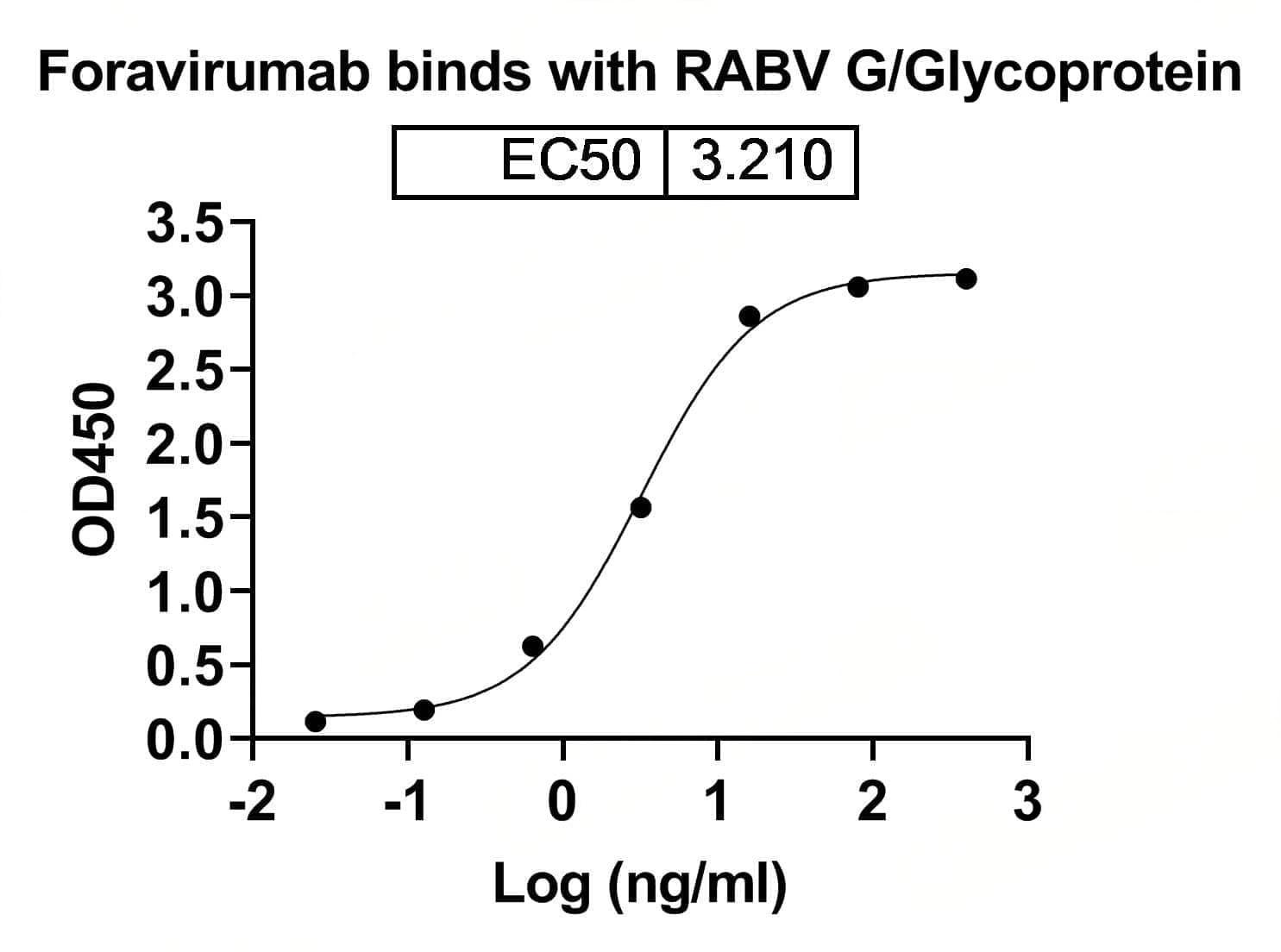
Bioactivity
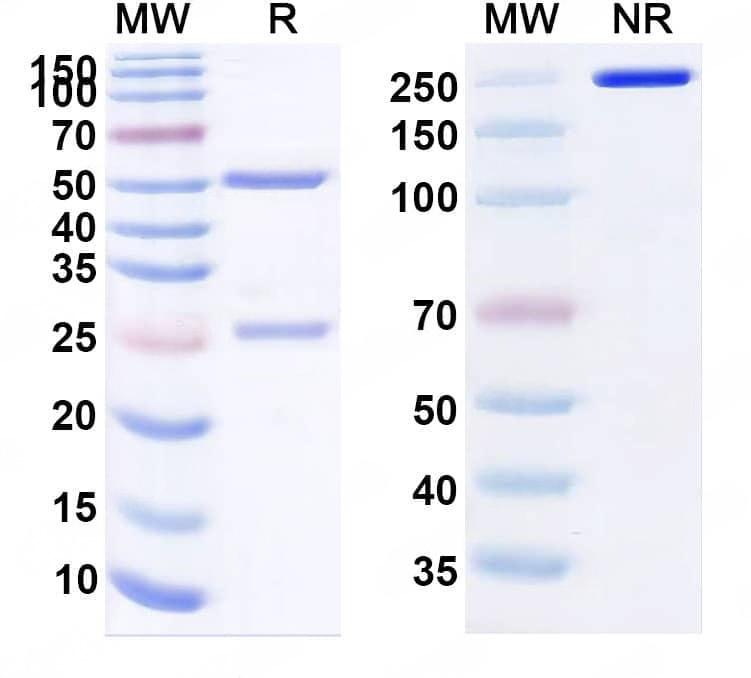
SDS-PAGE



- Catalog: TD-VK632016
- Clonality: Monoclonal
- Application: ELISA, Bioactivity: FACS, Functional assay, Research in vivo
- Synonyms: CR4098, 944548-38-3
-
规格:
询价
-
价格:
¥100ug
-
Catalog No.TD-VK632016
-
DescriptionForavirumab(TD-VK632016) is a research-grade recombinant antibody targetingG/Glycoprotein. Produced in mammalian cells with native-like glycosylation.HighlightsResearch Grade— For PK/PD studies, assay development, and ADA research.Native Glycosylation— Mammalian expression ensures native-like patterns.
-
Species reactivityRabies virus (RABV)
-
ApplicationsELISA, Bioactivity: FACS, Functional assay, Research in vivo
-
Host speciesHuman
-
IsotypeIgG1, kappa
-
ClonalityMonoclonal
-
TargetG/Glycoprotein
-
Endotoxin levelPlease contact the lab for this information.
-
Purity>95% purity as determined by SDS-PAGE.
-
PurificationProtein A/G purified from cell culture supernatant.
-
AccessionO92284
-
FormLiquid
-
Storage buffer0.01M PBS, pH 7.4.Please refer to the specific buffer information in the hardcopy of datasheet or the lot-specific COA.
-
Stability and StorageUse a manual defrost freezer and avoid repeated freeze-thaw cycles. Store at 4°C for short-term storage (1-2 weeks). Store at -20°C for up to 12 months. For long-term storage, store at -80°C.
-
Alternate NamesCR4098, 944548-38-3
-
BackgroundForavirumab (CR4098), is currently under phase II clinical trial in the form of a cocktail with rafivirumab (CR57), a mAb derived from somatic cell hybridization targeting rabies virus glycoprotein antigenic site I.• Structure and function of respiratory syncytial virus surface glycoproteins., PMID:24362685• Full-length G glycoprotein directly extracted from rabies virus with detergent and then stabilized by amphipols in liquid and freeze-dried forms., PMID:34297405• Recombinant Soluble Henipavirus Glycoprotein Preparation., PMID:37610572• Ephedrae Herba and Cinnamomi Cortex interactions with G glycoprotein inhibit respiratory syncytial virus infectivity., PMID:35079103• VSV-G Viral Envelope Glycoprotein Prepared fromPichia pastorisEnhances Transfection of DNA into Animal Cells., PMID:28297751• Spatiotemporal transcriptomic mapping of regenerative inflammation in skeletal muscle reveals a dynamic multilayered tissue architecture., PMID:39190487• Vesicular Stomatitis Virus (VSV) G Glycoprotein Can Be Modified to Create a Her2/Neu-Targeted VSV That Eliminates Large Implanted Mammary Tumors., PMID:37199666• Evolutionary trends of respiratory syncytial viruses: Insights from large-scale surveillance and molecular dynamics of G glycoprotein., PMID:38784562• G glycoprotein amino acid residues required for human monoclonal antibody RAB1 neutralization are conserved in rabies virus street isolates., PMID:21693135• The use of monoclonal antibodies and lectins to identify changes in viral glycoproteins that are influenced by glycosylation: the case of human respiratory syncytial virus attachment (G) glycoprotein., PMID:17502674
-
CaptionSEC-HPLC detection for Research Grade Foravirumab. | Detects Human G/Glycoprotein in indirect ELISAs. | SDS-PAGE for Research Grade Foravirumab.
-
NoteFor research use only. Not suitable for clinical or therapeutic use.
