Research Grade Gontivimab
All
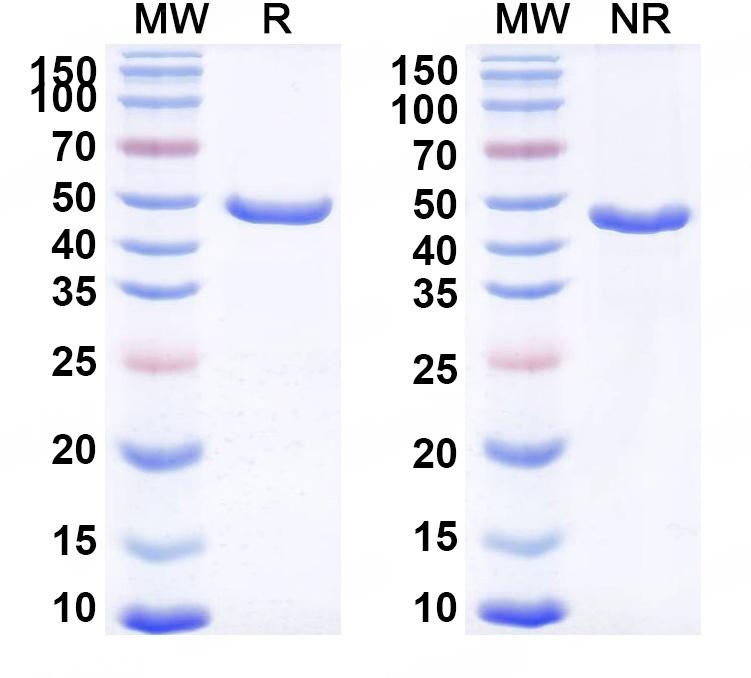
SDS-PAGE

- Catalog: TD-VK421066
- Clonality: Monoclonal
- Application: ELISA, Bioactivity: FACS, Functional assay, Research in vivo
- Synonyms: ALX-0171, VR-465, 1257358-38-5
-
规格:
询价
-
价格:
¥100ug
-
Catalog No.TD-VK421066
-
DescriptionGontivimab(TD-VK421066) is a research-grade recombinant antibody targetingF. Produced in mammalian cells with native-like glycosylation.HighlightsResearch Grade— For PK/PD studies, assay development, and ADA research.Native Glycosylation— Mammalian expression ensures native-like patterns.
-
Species reactivityHRSV-A
-
ApplicationsELISA, Bioactivity: FACS, Functional assay, Research in vivo
-
Host speciesLama/glama
-
IsotypeVH-VH-VH
-
ClonalityMonoclonal
-
TargetF, Fusion glycoprotein F0, Fusion glycoprotein F2, p27, Intervening segment, Pep27, Peptide 27, Fusion glycoprotein F1
-
Endotoxin levelPlease contact the lab for this information.
-
Purity>95% purity as determined by SDS-PAGE.
-
PurificationProtein A/G purified from cell culture supernatant.
-
AccessionP03420
-
FormLiquid
-
Storage buffer0.01M PBS, pH 7.4.Please refer to the specific buffer information in the hardcopy of datasheet or the lot-specific COA.
-
Stability and StorageUse a manual defrost freezer and avoid repeated freeze-thaw cycles. Store at 4°C for short-term storage (1-2 weeks). Store at -20°C for up to 12 months. For long-term storage, store at -80°C.
-
Alternate NamesALX-0171, VR-465, 1257358-38-5
-
BackgroundGontivimab is a fully human IgG monoclonal antibody (mAb) developed using recombinant DNA technology for the prevention of respiratory syncytial virus (RSV) infections. It is designed to provide passive immunization to individuals at elevated risk of severe disease, particularly infants with prematurity or underlying medical conditions. Gontivimab is directed against a conserved neutralizing epitope within the prefusion conformation of the RSV Fusion (F) protein, a structural state critical for viral membrane fusion and host-cell entry. By targeting this highly accessible antigenic site, Gontivimab achieves potent inhibition of both RSV-A and RSV-B strains.Gontivimab is composed entirely of human immunoglobulin gene sequences without murine elements. The heavy chain derives from human IgG1 constant domains and variable region frameworks from VH gene families commonly involved in high-affinity antiviral responses. The light chain contains the human Cκ constant domain and kappa variable region frameworks. Antigen specificity is conferred by complementarity-determining regions selected to recognize the conserved prefusion F epitope with high affinity and stability. As with other fully human antibodies, this design minimizes immunogenicity while maintaining strong neutralizing activity.Structurally, Gontivimab consists of two heavy chains and two light chains, forming a typical IgG molecule with an approximate molecular weight of 145,000–150,000 Daltons. Preclinical studies show that Gontivimab markedly reduces viral replication, airway inflammation, and lower-respiratory tract pathology. Fc engineering has been applied to extend serum half-life, allowing prolonged protective levels throughout the RSV season with fewer injections.Although still in clinical development, Gontivimab is intended for prophylaxis in infants and other vulnerable groups at higher risk of severe RSV disease, including those with bronchopulmonary dysplasia or chronic lung disease, and patients with hemodynamically significant congenital heart disease. These populations experience greater morbidity, hospitalization frequency, and long-term respiratory complications following RSV infection.Through its fully human sequence, targeted epitope specificity, and extended half-life, Gontivimab represents a next-generation monoclonal antibody for RSV prevention and a valuable molecule for advancing research on viral neutralization, antibody engineering, and RSV immunopathogenesis.
-
CaptionSDS-PAGE for Research Grade Gontivimab
-
NoteFor research use only. Not suitable for clinical or therapeutic use.
