Research Grade Polatuzumab
All
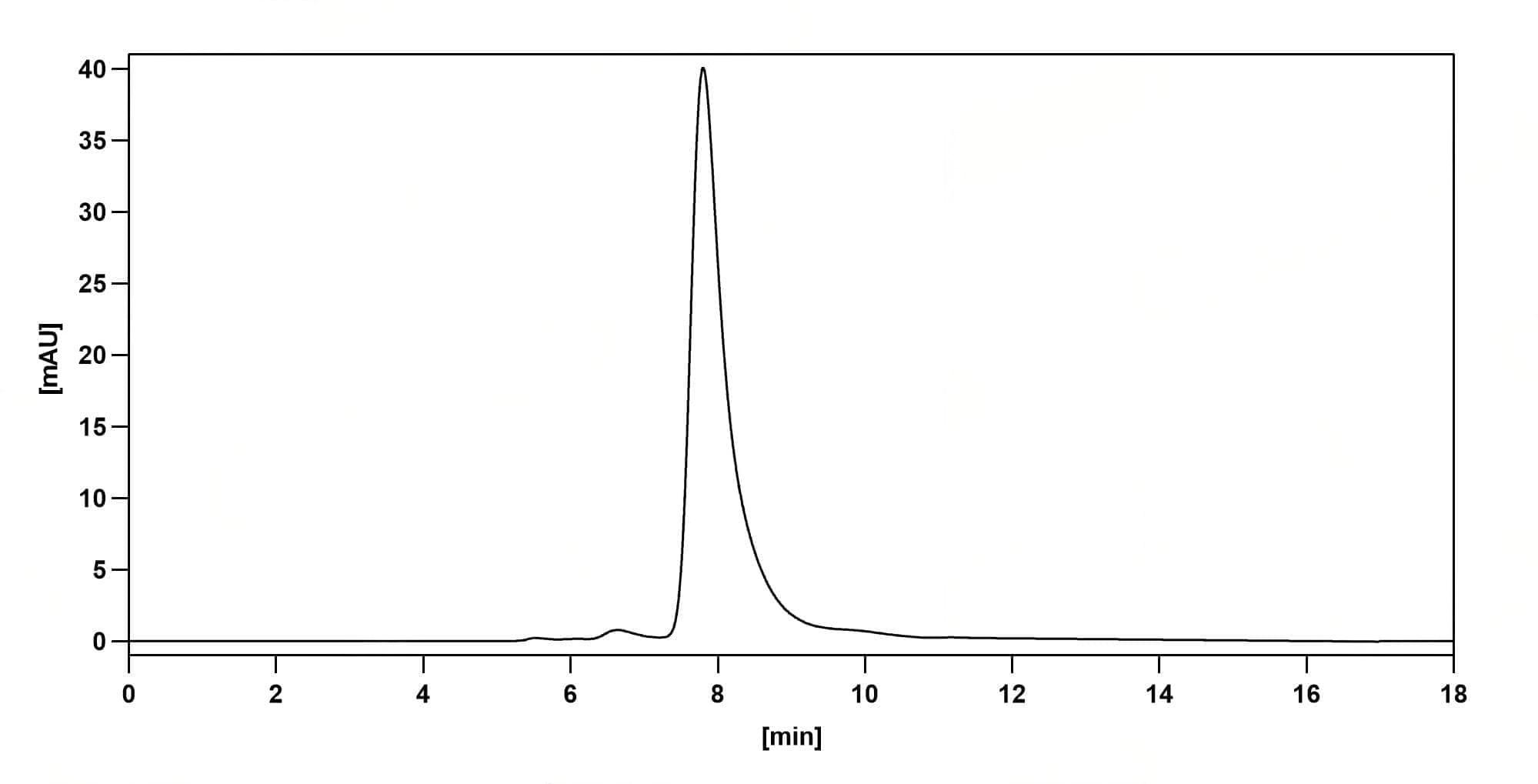
SEC-HPLC
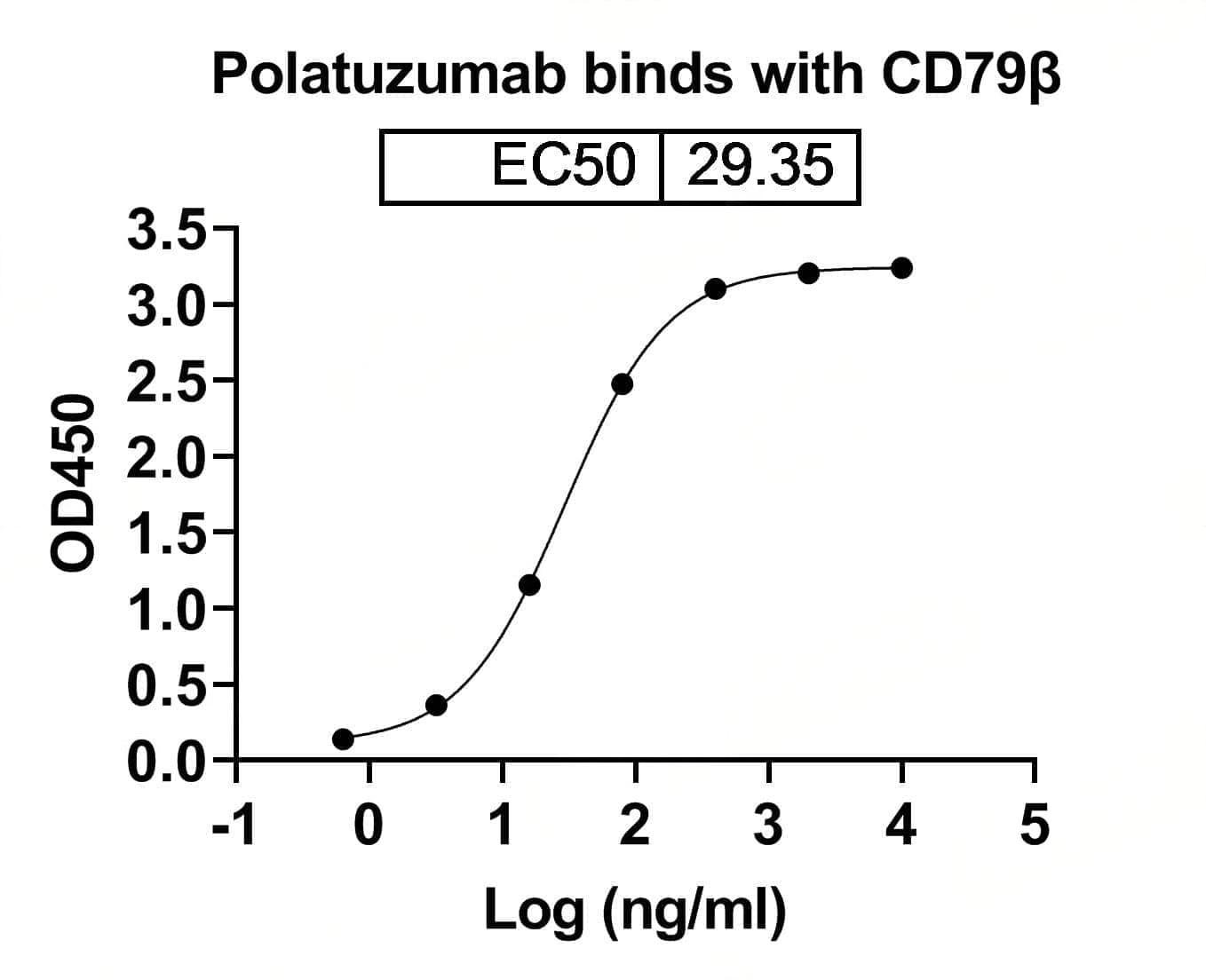
Bioactivity
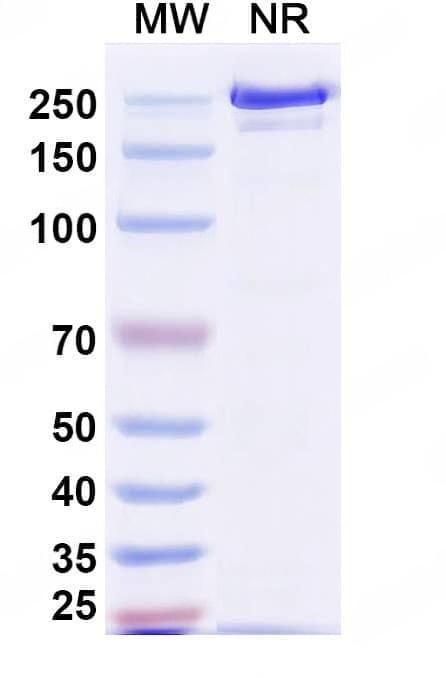
SDS-PAGE



- Catalog: TD-HW431016
- Clonality: Monoclonal
- Application: ELISA, Bioactivity: FACS, Functional assay, Research in vivo
- Synonyms: ACD79B-VCMMAE, DCDS4501A, FCU2711, RO5541077-000, polatuzumabvedotin-piiq, 1313206-42-6
-
规格:
询价
-
价格:
¥100ug
-
Catalog No.TD-HW431016
-
DescriptionPolatuzumab(TD-HW431016) is a research-grade recombinant antibody targetingCD79B. Produced in mammalian cells with native-like glycosylation.HighlightsResearch Grade— For PK/PD studies, assay development, and ADA research.Native Glycosylation— Mammalian expression ensures native-like patterns.
-
Species reactivityHuman
-
ApplicationsELISA, Bioactivity: FACS, Functional assay, Research in vivo
-
Host speciesHumanized
-
IsotypeIgG1-kappa
-
ClonalityMonoclonal
-
TargetB-cell-specific glycoprotein B29, CD79B, B-cell antigen receptor complex-associated protein beta chain, Ig-beta, B29, Immunoglobulin-associated B29 protein, IGB, CD79b
-
Endotoxin levelPlease contact the lab for this information.
-
Purity>95% purity as determined by SDS-PAGE.
-
PurificationProtein A/G purified from cell culture supernatant.
-
AccessionP40259
-
FormLiquid
-
Storage buffer0.01M PBS, pH 7.4.Please refer to the specific buffer information in the hardcopy of datasheet or the lot-specific COA.
-
Stability and StorageUse a manual defrost freezer and avoid repeated freeze-thaw cycles. Store at 4°C for short-term storage (1-2 weeks). Store at -20°C for up to 12 months. For long-term storage, store at -80°C.
-
Alternate NamesACD79B-VCMMAE, DCDS4501A, FCU2711, RO5541077-000, polatuzumabvedotin-piiq, 1313206-42-6
-
BackgroundPolatuzumab vedotin is an antibody-drug conjugate formed by a CD79b antibody conjugated to the highly cytotoxic agent monomethyl auristatin E by means of a cleavable linker. Following significant clinical efficacy in R/R DLBCL, polatuzumab vedotin was granted accelerated Food and Drug Administration (FDA) approval in combination with bendamustine plus rituximab for patients who have failed at least two prior therapies. Other clinical studies involving polatuzumab vedotin in combination with other therapy regimens are also under evaluation for previously untreated DLBCL patients.
-
CaptionSEC-HPLC detection for Research Grade Polatuzumab. | Detects Human CD79β/CD79B in indirect ELISAs. | SDS-PAGE for Research Grade Polatuzumab.
-
NoteFor research use only. Not suitable for clinical or therapeutic use.
