Research Grade Pacmilimab
All
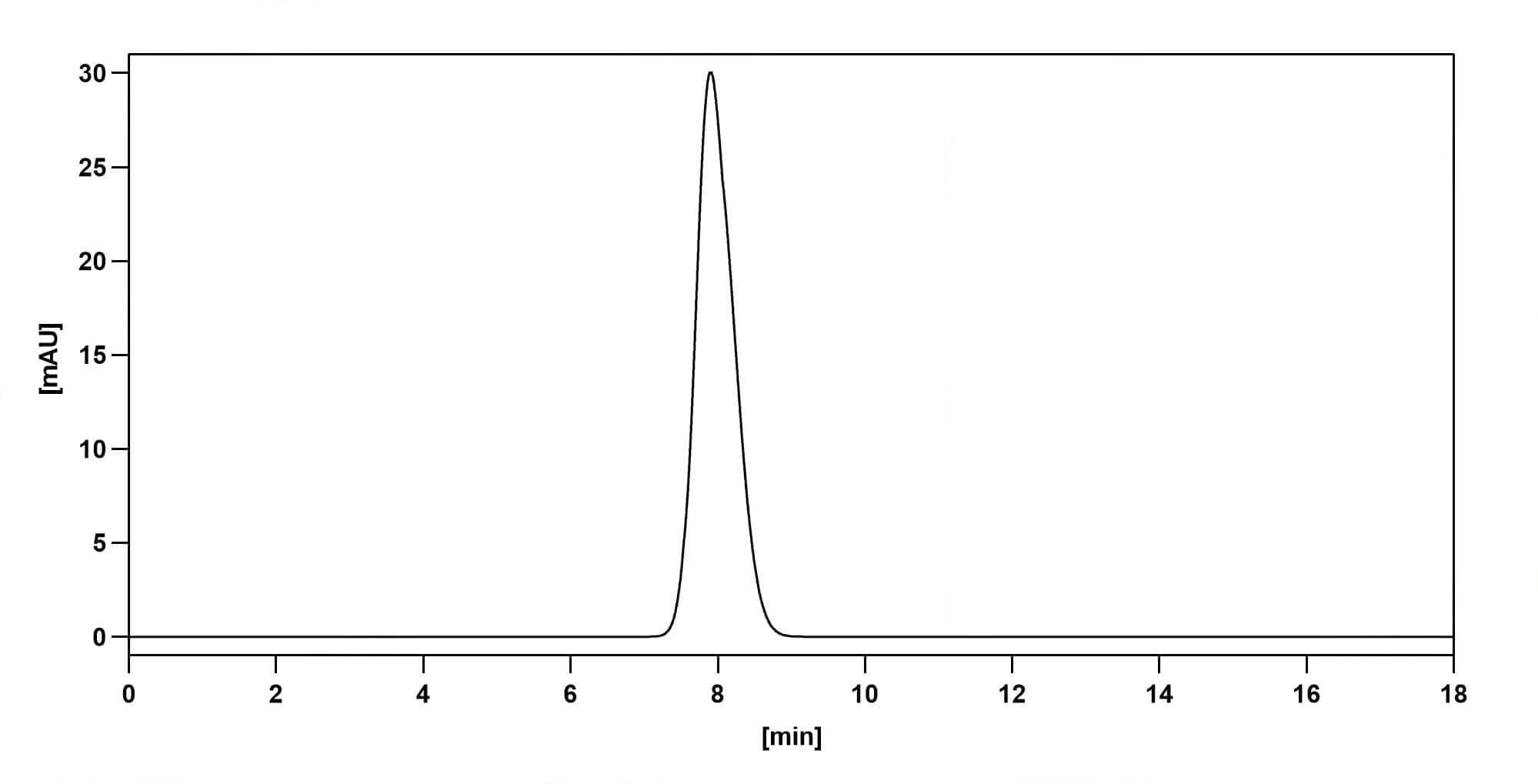
SEC-HPLC
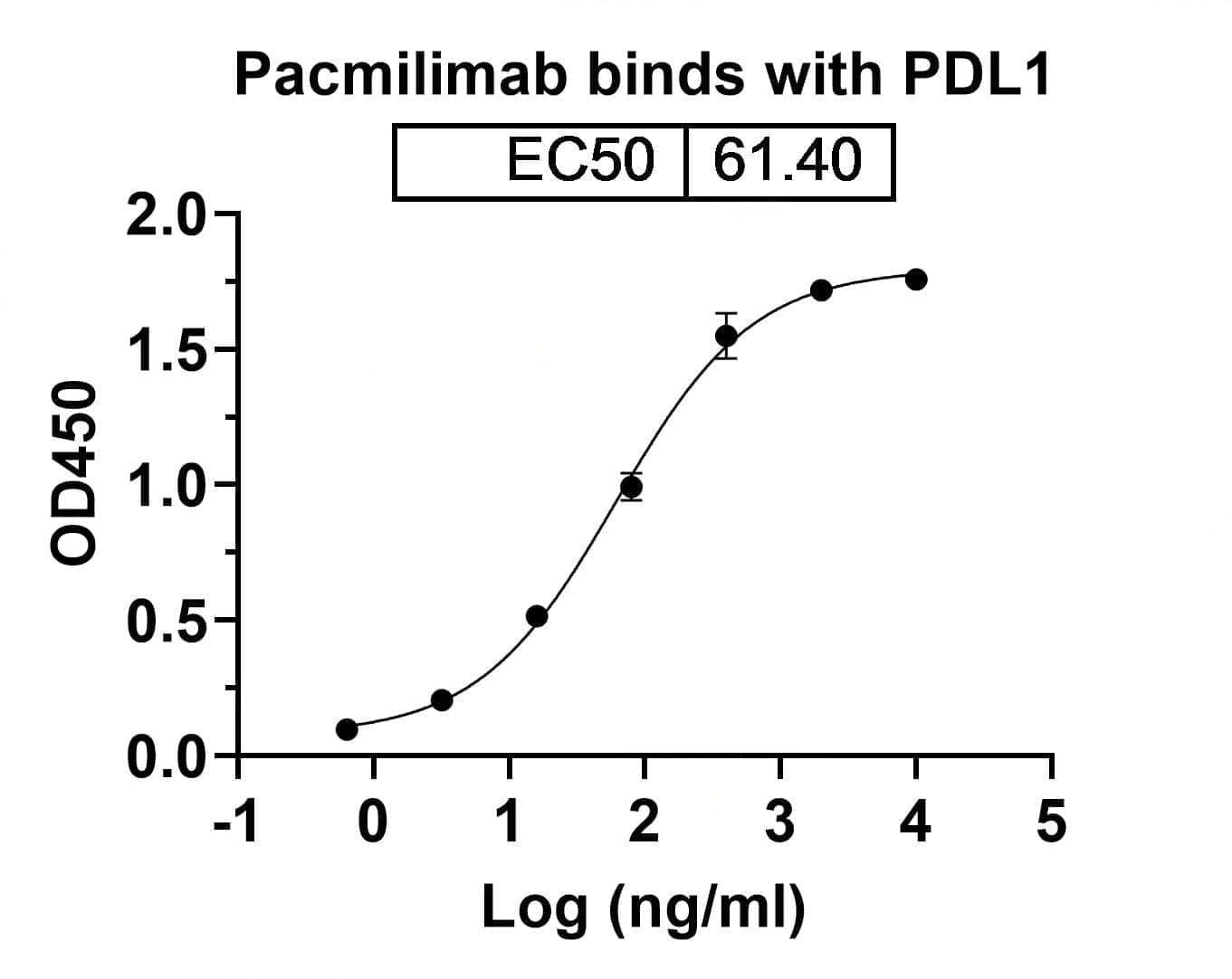
Bioactivity
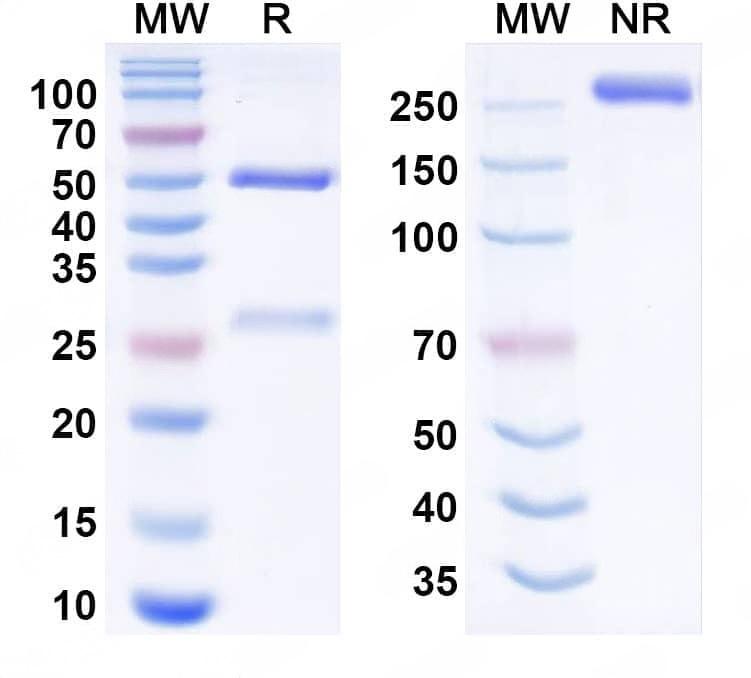
SDS-PAGE




- Catalog: TD-HV974056
- Clonality: Monoclonal
- Application: ELISA, Bioactivity: FACS, Functional assay, Research in vivo
- Synonyms: CX-072, 2145091-51-4
-
规格:
询价
-
价格:
¥100ug
-
Catalog No.TD-HV974056
-
DescriptionPacmilimab(TD-HV974056) is a research-grade recombinant antibody targetingCD1. Produced in mammalian cells with native-like glycosylation.HighlightsResearch Grade— For PK/PD studies, assay development, and ADA research.Native Glycosylation— Mammalian expression ensures native-like patterns.
-
Species reactivityHuman
-
ApplicationsELISA, Bioactivity: FACS, Functional assay, Research in vivo
-
Host speciesHuman
-
IsotypeIgG4-kappa
-
ClonalityMonoclonal
-
TargetB7-H1, Programmed cell death 1 ligand 1, PDCD1 ligand 1, PDCD1L1, B7 homolog 1, PDCD1LG1, PDL1, hPD-L1, Programmed death ligand 1, B7H1, PD-L1, CD274
-
Endotoxin levelPlease contact the lab for this information.
-
Purity>95% purity as determined by SDS-PAGE.
-
PurificationProtein A/G purified from cell culture supernatant.
-
AccessionQ9NZQ7
-
FormLiquid
-
Storage buffer0.01M PBS, pH 7.4.Please refer to the specific buffer information in the hardcopy of datasheet or the lot-specific COA.
-
Stability and StorageUse a manual defrost freezer and avoid repeated freeze-thaw cycles. Store at 4°C for short-term storage (1-2 weeks). Store at -20°C for up to 12 months. For long-term storage, store at -80°C.
-
Alternate NamesCX-072, 2145091-51-4
-
BackgroundAtezolizumab (trade name Tecentriq) is a fully humanized, engineered monoclonal antibody of IgG1 isotype against the protein programmed cell death-ligand 1 (PD-L1). In 2015, it was in clinical trials as an immunotherapy for several types of solid tumors. It was under investigation by Genetech/Roche. In April 2016, Roche announced that atezolizumab had been granted fast track status for lung cancer by the FDA. In May 2018, Tecentriq was in combination with Avastin and standard chemotherapy for some patients with lunch cancer was granted priority review.
-
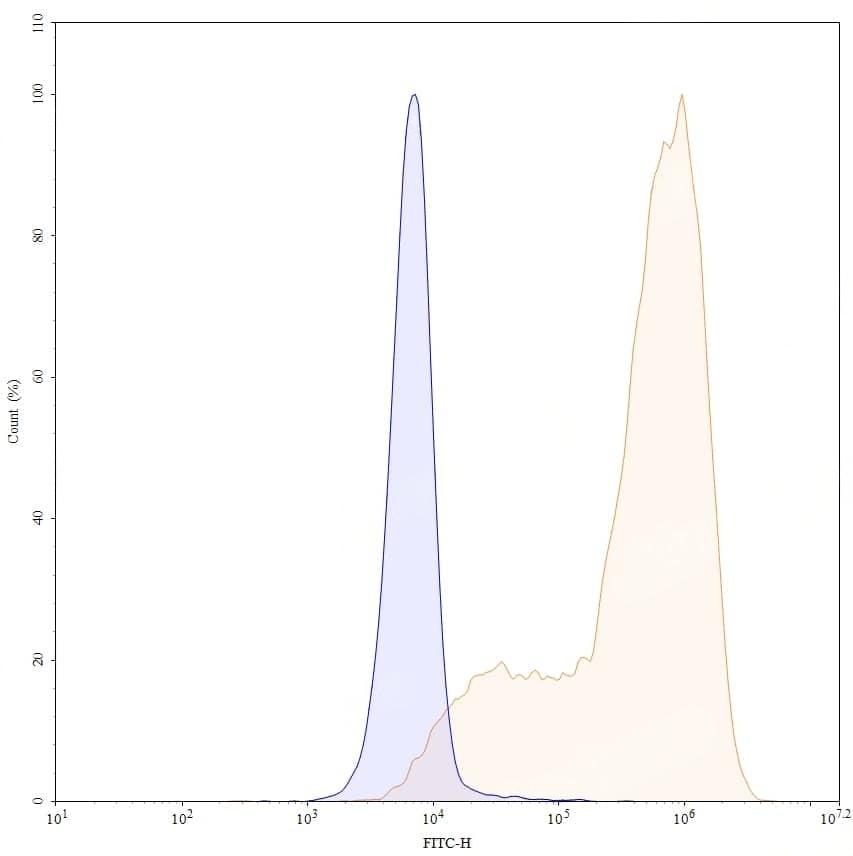
CaptionSEC-HPLC detection for Research Grade Pacmilimab. | Detects Human CD274/PD-L1/B7-H1 in indirect ELISAs. | SDS-PAGE for Research Grade Pacmilimab. | Flow-cytometry using anti-human CD274 antibody. Untransfected cells (blue Histogram) and Transfected cells (Yellow Histogram) were stained with an anti-human CD274 monoclonal antibody (Catalog: HV974056) at a concentration of 5 µg/ml for 30 mins at RT. After washing, bound antibody was detected using a Goat Anti-Human IgG H&L Polyclonal Antibody, FITC (abinScience: HF690414) and cells analysed on a NovoCyte Flow Cytometer.
-
NoteFor research use only. Not suitable for clinical or therapeutic use.
