Research Grade Sabatolimab
All
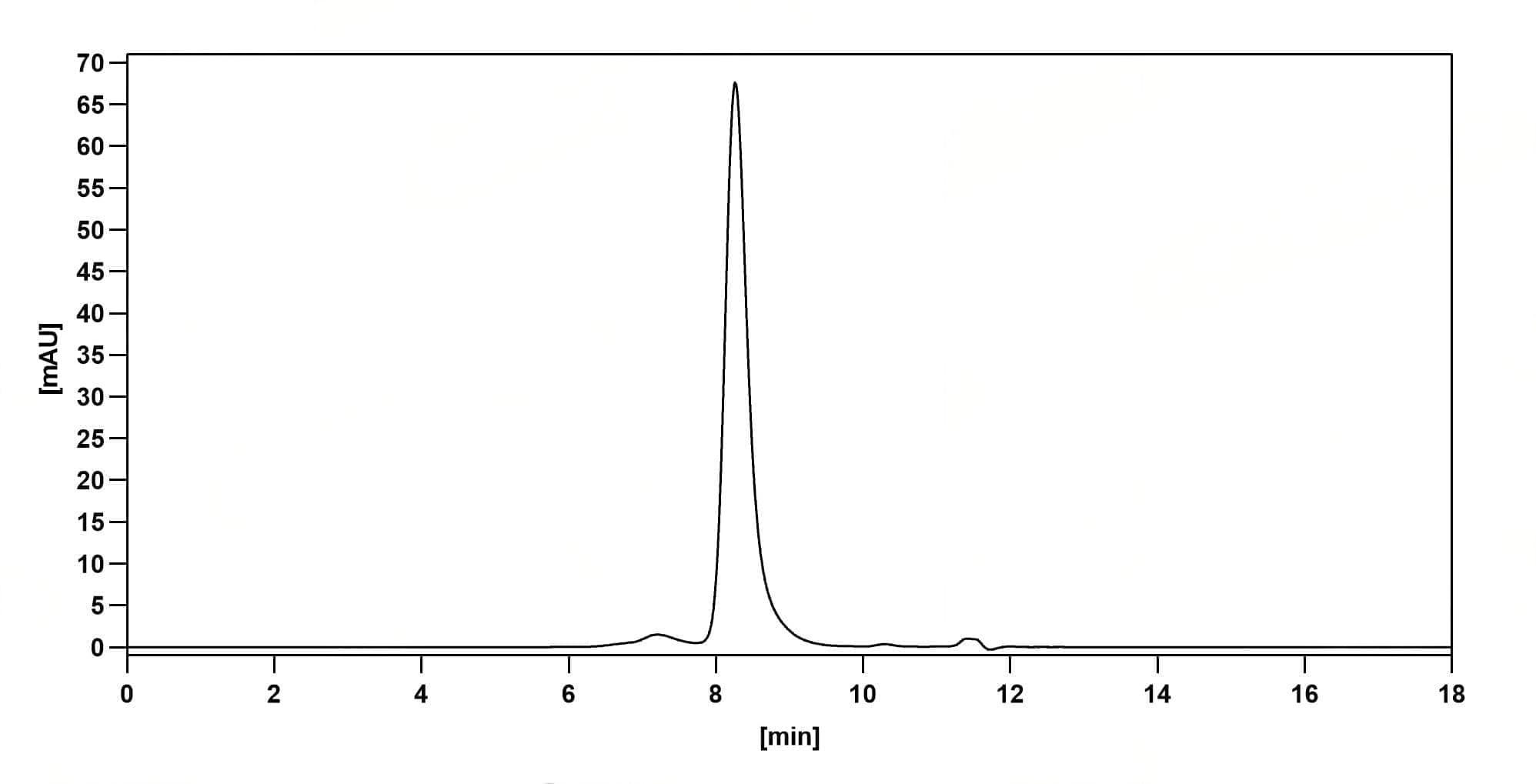
Bioactivity
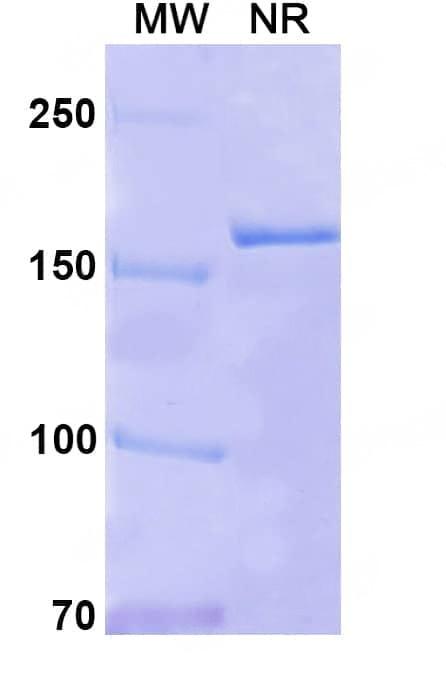
SDS-PAGE


- Catalog: TD-HV029026
- Clonality: Monoclonal
- Application: ELISA, Bioactivity: FACS, Functional assay, Research in vivo
- Synonyms: MBG-453, 2252262-24-9
-
规格:
询价
-
价格:
¥100ug
-
Catalog No.TD-HV029026
-
DescriptionSabatolimab(TD-HV029026) is a research-grade recombinant antibody targetingCD366. Produced in mammalian cells with native-like glycosylation.HighlightsResearch Grade— For PK/PD studies, assay development, and ADA research.Native Glycosylation— Mammalian expression ensures native-like patterns.
-
Species reactivityHuman
-
ApplicationsELISA, Bioactivity: FACS, Functional assay, Research in vivo
-
Host speciesHumanized
-
IsotypeIgG4-kappa
-
ClonalityMonoclonal
-
TargetT-cell immunoglobulin mucin receptor 3, HAVcr-2, TIMD-3, T-cell membrane protein 3, HAVCR2, Hepatitis A virus cellular receptor 2, CD366, TIMD3, TIM-3, TIM3, T-cell immunoglobulin and mucin domain-containing protein 3
-
Endotoxin levelPlease contact the lab for this information.
-
Purity>95% purity as determined by SDS-PAGE.
-
PurificationProtein A/G purified from cell culture supernatant.
-
AccessionQ8TDQ0
-
FormLiquid
-
Storage buffer0.01M PBS, pH 7.4.Please refer to the specific buffer information in the hardcopy of datasheet or the lot-specific COA.
-
Stability and StorageUse a manual defrost freezer and avoid repeated freeze-thaw cycles. Store at 4°C for short-term storage (1-2 weeks). Store at -20°C for up to 12 months. For long-term storage, store at -80°C.
-
Alternate NamesMBG-453, 2252262-24-9
-
BackgroundVerzistobart is an IgG1κ antibody targeting the hepatitis A virus cellular receptor HAVCR2.
-
CaptionSEC-HPLC detection for Research Grade Sabatolimab. | SDS-PAGE for Research Grade Sabatolimab
-
NoteFor research use only. Not suitable for clinical or therapeutic use.
